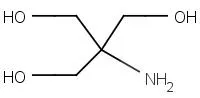
Product Description
- Chemical Name: tris (hydroxymethyl) aminomethane
- English Name: Trometamol
- Abbreviation: Tris
- CAS#: 77-86-1
- Molecular Weight: 121.135
- Formula: C4H11NO3
- Storage: Room temperature, away from light and moisture
- Appearance: White crystal powder
Application: The product can be used for the preparation of buffer solution in biochemistry and molecular biology experiments, pharmaceutical intermediates, surfactants and curing accelerators, water-soluble polymers containing Tris structure units as paint dispersants, and formaldehyde adsorption materials.
| CAS Number | 1185-53-1 |
| Molecular Formula | C4H12ClNO3 |
| Molecular Weight | 157.6 |
| EINECS | 214-684-5 |
| Appearance | White Powder |
| Melting Point | 150-152 °C |
| Storage Conditions | Room Temp |
Configuration Method of Tris Solution
1
Weigh the required Tris powder and add to sterile high purity water.
2
Use a magnetic stirrer to fully dissolve until completely homogeneous.
3
Concentration adjustment: Generally 1 mol/L, adjustable based on specific needs.
4
Measure and control PH value between 7.0-8.5 using a PH meter.
5
Standardize the solution volume, typically used at 50-200 mmol/L for buffering.
6
Sterilize the solution in sterile bottles according to standard procedures.
Safety Note: Strictly observe chemical safety procedures. Wear personal protective equipment to avoid skin/eye contact, inhalation, or ingestion. Check labels and production dates to avoid expired chemicals.
Frequently Asked Questions
What are the primary applications of Tris powder?
Tris is widely used as a biological buffer for DNA/RNA extraction and cell culture, a chemical raw material for coatings, and a cross-linking agent for resins.
What is the purity level of the Tris HCL product?
The product features high purity, maintained at above 99%, ensuring reliable results in laboratory and industrial applications.
What are the recommended storage conditions for Tris?
It should be stored at room temperature in a dry place, kept away from direct light and moisture to prevent degradation.
How is the PH value controlled during solution preparation?
The PH value should be monitored using a calibrated PH meter and is generally controlled within the range of 7.0 to 8.5 for standard buffer solutions.
Is Tris HCL soluble in water?
Yes, Tris HCL is highly soluble in water, making it ideal for creating homogeneous buffer solutions for molecular biology.
What safety equipment should be used when handling this chemical?
Personal protective equipment, including gloves and safety goggles, should be worn to avoid skin contact, eye irritation, or accidental inhalation.